Bridges for Pharma and Medical Devices
One click information access for improved development and delivery of solutions to improve our health
Overview
In the Pharmaceuticals and Medical Devices industry, access to relevant and up-to-date information is critical to ensure efficient and effective operations. However, workers often navigate through multiple systems to gather the necessary data, interrupting their workflow and reducing productivity. With Bridges, workers can access the information they need without leaving their current task, allowing them to maintain focus and efficiency. Whether it’s reviewing product specifications, or accessing regulatory information, Bridges streamlines the information search process and empowers workers to make informed decisions.
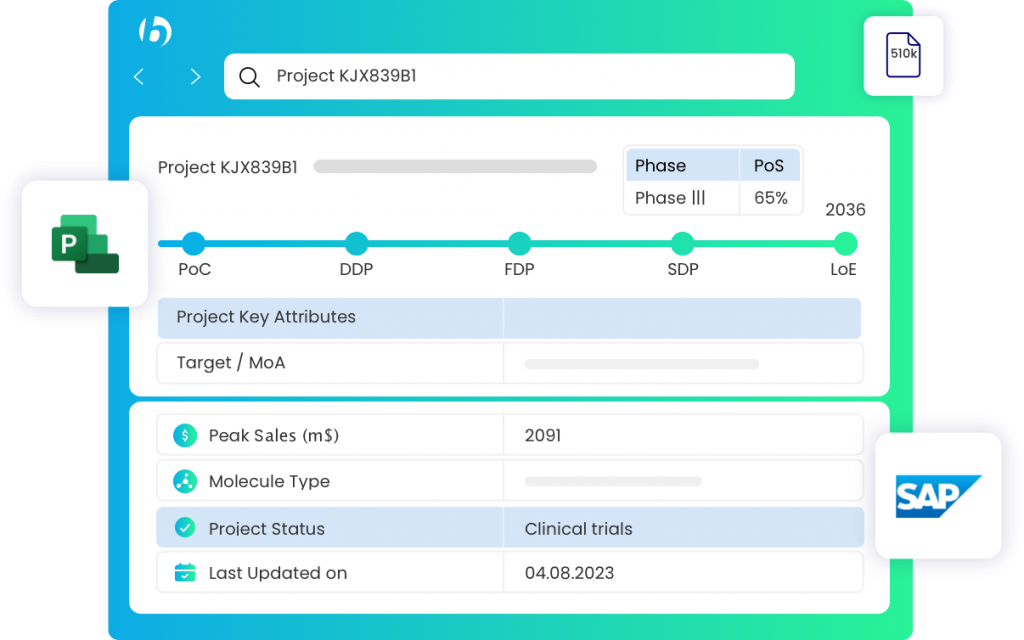
How can Bridges assist
Clinical R&D
While designing clinical trials, analyzing data, and reporting results, researchers can harness Bridges to quickly retrieve data from the pharma development projects, see project status, next approval steps and access project documents.
Regulatory Affairs
For tasks like ensuring drugs compliance, preparing regulatory filings, and monitoring changes in regulatory requirements, Bridges offers one click access to documents management system and PLM.
Engineering
Engineers developing a new device can use Bridges for rapid access of data to an integrated view of PLM and ERP systems.
Sales
Medical devices salespersons will improve their productivity, while preparing a customer quote, if they uses Bridges for a unified view of relevant ERP and CRM systems.
Our Value Proposition
Critical Information on Desktop
Information Integration
Critical Information on Desktop
important information from various enterprise applications is being seamlessly incorporated into the user’s desktop, making it easier for the user to access and utilize the information in a mission-critical manner.
Information Integration
Facilitating seamless integration between disparate information sources through a user-friendly approach
Testimonials

“Bridges is a must have if you are tired of using Agile for tedious item look-up”

“Great if not the greatest search tool”
